Certification opens up market opportunities - Neogen
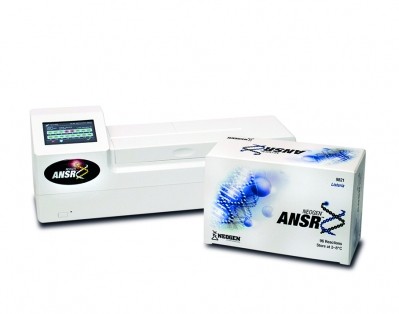
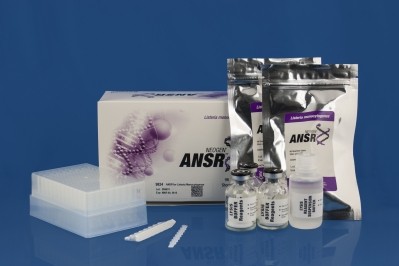
The firm’s ANSR for Salmonella was validated and approved with the criteria of a NF Validation study carried out according to the EN ISO 16140 standard.
It uses an isothermal DNA amplification process and fluorescent molecular beacon technology for detection of the pathogen target.
The validation (NF Validation by AFNOR certification NEO 35/02-0513) follows the kit’s approval by AOAC International.
“The NF validation of ANSR for Salmonella will allow Neogen access to a number of European markets, and beyond, where the use of the test requires an accreditation study carried out according to the AFNOR standard,” said Neogen’s Steve Chambers.
Neogen said the test cuts the wait time for definitive results, with only 10 minutes of reaction time following sample enrichment, compared to other commercially available molecular amplification tests which require up to three hours of reaction time.
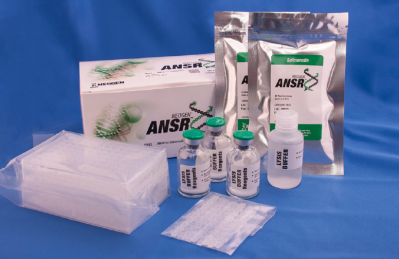
The firm is also pursuing a similar validation of its ANSR for Listeria rapid molecular test system.