Microbes – including bacteria, yeasts, algae and fungi – are being used in the production of a growing number of foods. But what happens to those microbes after use?
With consumers increasingly aware of the health consequences of their diet and businesses facing an increasingly challenging economic environment, is there a way to repurpose leftover microbes to help both groups? Microbiology expert Joyce Mulder and gut health and immunity expert Torsten Scheithauer, both from NIZO, discuss the potential health benefits of extracting fibre from microbial side streams.
René Floris: Why should the industry be interested in fibre from microbial side streams?
Torsten Scheithauer: There is a lot of evidence to suggest that Western diets tend to be low in fibre.
Although human bodies can’t digest fibre, many of the microbes that live in our gut can. So, eating enough of the right kinds of fibre can promote healthy microbiota which then produce helpful metabolites such as short-chain fatty acids. There are well established links in the literature between low dietary fibre levels and health issues such as obesity, diabetes and inflammatory (bowel) diseases.
Joyce Mulder: At the same time, more and more food production processes are turning to microbes such as bacteria, yeasts, algae and fungi – this includes traditional fermentation-based industries like brewing but also newer applications like kombucha or even using microbes to create protein. Once it has been used for its initial purpose, most of this microbial biomass is currently considered as waste. But if we could extract the fibres from the microbes, we would have a highly sustainable way of increasing fibre in the diet and boosting gut and overall health. And by making fuller use of an otherwise waste material, it could also open up new revenue opportunities for companies already using microbes.
RF: How does fibre promote health?
JM: Fibres can be fermented by beneficial bacteria, keeping a healthy balance in the gut between these and organisms that could cause illness under certain conditions. When digesting the fibre, the beneficial bacteria produce compounds that are useful to our bodies. For instance, these metabolites can be beneficial to our gut (barrier) function, immune response and microbiota composition / functionality.
TS: The concept of trained immunity is a good example. If we eat certain types of fibre, this can stimulate beneficial bacteria in our gut to produce metabolites that influence our immune system response to certain triggers in different ways. One type of fibre could prime our immune system so that it later responds more strongly when it encounters a pathogen, helping us prevent infections and perhaps even certain cancers. On the other hand, ingesting other fibres could lead to metabolites that induce a tolerance in the immune system, damping down the response to later triggers. This could improve overall wellbeing and particularly help those suffering from inflammatory diseases such as ulcerative colitis and Crohn’s disease, potentially alleviating some symptoms.
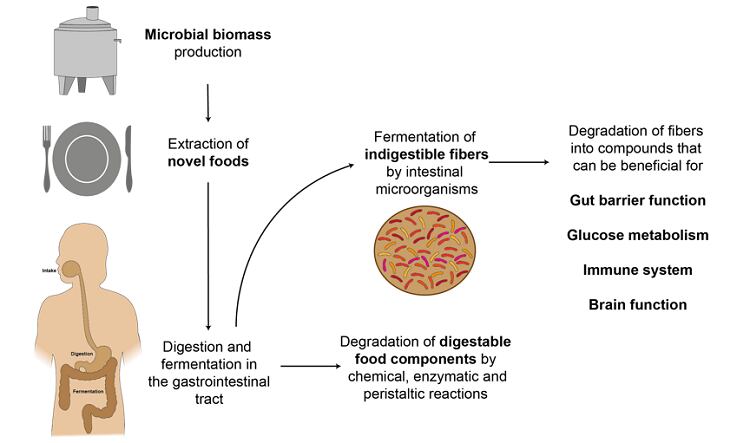
RF: What kind of fibres can be found in microbes?
TS: Probably the most familiar and most studied microbial fibres are the beta-glucans extracted from yeasts. These have already been shown to stimulate the immune system and to modulate gut microbiota composition. However, there is a great deal of structural variation between beta-glucans from different sources, which can lead to very different effects on the body. For example, beta-glucans from oats don’t stimulate the immune system in the same way as yeast beat-glucans, but are known to promote glycaemic control and reduce cholesterol levels.
JM: All microorganisms contain various types of fibre. So far, the food industry has focused, perhaps understandably, on the protein content of microbes and the fibre content has been overlooked. When you consider all the different bacteria, fungi, yeasts and algae used in food production, science has so far explored a small fraction of the possibilities of exploiting those fibres. There is a huge uncharted landscape in terms of the range of microbes and the individual types of fibre. We believe there are huge opportunities for the industry to broaden its view and explore this landscape.
RF: How can the industry start exploring this area more extensively?
JM: We would like to see a much more thorough investigation of the fibre content of microbes and their potential health benefits. That requires collaboration from numerous industry players: food producers that have whole microbial biomass or access to microbial side
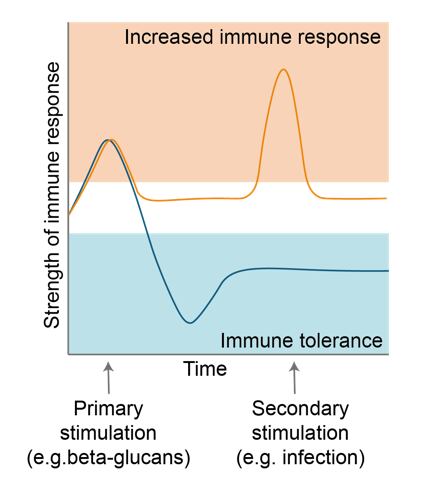
streams, organisations with experience in extracting fractions from microbes and experts in identifying the health benefits of specific nutritional elements.
TS: To stimulate that cooperation, NIZO is currently developing an application for funding to the Dutch government’s Top Sector Agri & Food initiative for a research project that adds value to microbial biomass by assessing potential health benefits of microbe-derived fibres. The planned project aims to bring together all the key players to explore both the health benefits for individuals and the commercial opportunities for businesses. If successful, the project will start in early 2024, and we are currently recruiting interested partners.
RF: What is the scope of the consortium?
JM: The project aims to identify promising candidates for health-promoting fibre ingredients derived from existing commercial streams of microbes – potentially laying the groundwork for designing clinical studies. That starts by looking at the biomass / stream and the relevant organisms it contains, then working out how best to extract the fibre fractions in each specific case. Extraction is again an area where the focus has so far been on proteins, but there have been some notable successes in fibre extraction as well, such as the EGGcited consortium, which recently announced a semi-industrial process for extracting fibre from brewers spent yeast.
TS: The next step is to characterise the extracted fibres by testing parameters such as viscosity, solubility and ability to absorb toxins. Then we explore the potential health benefits of the fibre through in vitro studies using a pipeline developed by NIZO. This involves putting the fibre (or whole biomass e.g. intact microbial cells) through in vitro digestion models and then introducing the output to intestinal and / or immune cells to see if there is any beneficial effect such as modulating cytokine production. The final step of the pipeline uses NIZO’s MicroColon to explore any changes the fibre induces in the composition of the gut microbiome and the metabolites it produces. These metabolites can also be investigated using intestinal and immune cell models to explore the subsequent effects of the fibre fermentation in the gut onto the gut health and immunity. If these initial investigations were promising, we would then look into the safety and regulatory requirements for later in vivo studies and potential market introductions.
JM: Of course, any products based on fibres extracted from as-yet unexplored microbial sides streams are a long way off. But by broadening the industry’s perspective of what the microbes we already use in food production could do for us, the project could be a significant first steppingstone towards new business opportunities for food producers and major health gains for individuals and at a societal level.
In NIZO's next article, the research organisation will look at the fermentation of hybrid food.